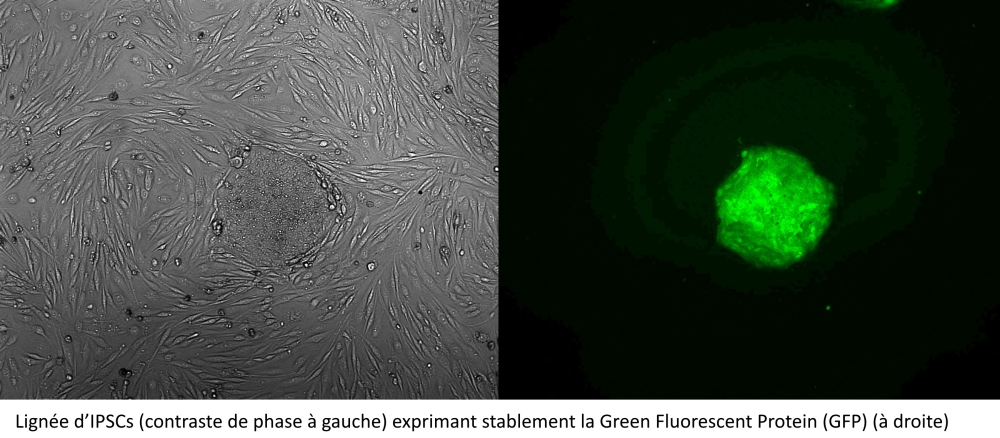
Genome editing
I-Stem’s CRISPR platform is dedicated to the production of genetically modified human pluripotent stem cell lines for the study and treatment of single gene disorders.
Since its discovery, the CRISPR-Cas9 genome editing technique has continued to evolve and offer new applications. On the platform, we use the CRISPR-Cas system (CASS) to modify the genome of human induced pluripotent stem cell lines (IPSCs) by indels creation (nucleotide insertion/deletions), base editing or homologous directed recombination (HDR) approaches.
The goal is to generate :
- IPSCs with heterozygous or homozygous loss of gene expression
- IPSCs “tool” lines to follow the expression of a gene or to induce its overexpression
- IPSCs lines carrying genetic mutations detected in a patient for whom no cell sample is available
- isogenic IPSCs lines that are controls of patient IPSCs lines in which the genetic mutation has been corrected.
The genetically modified IPSC clones produced by the platform are delivered after ON-TARGETS and OFF-TARGET tests of the CCAS system and quality control (absence of mycoplasma, maintenance of pluripotency and genomic integrity).
Team members
Sandrine Baghdoyan, PhD-HDR
Platform Manager - INSERM
Pascal Fragner, PhD
Platform Engineer CECS
Michel Cailleret
Engineer INSERM
Publications
Semi-automated optimized method to isolate CRISPR/Cas9 edited human pluripotent stem cell clones.
27 April 2023
Stem cell research & therapy
Pathological modeling of glycogen storage disease type III with CRISPR/Cas9 edited human pluripotent stem cells.
01 January 2023
Frontiers in cell and developmental biology
Team equipment

Electroporation system Neon
Editing of IPS lines.
This model has a tip chamber that generates a uniform electric field that greatly increases transfection efficiency and cell viability.
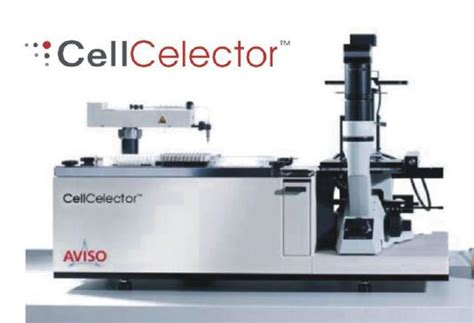
CellCelector
For high-throughput cloning of edited cells, we use this automatic sampling machine for single cells and colonies.

Bioanalyzer Agilent 2100
For evaluation of clone editing, we use this automated electrophoresis system.
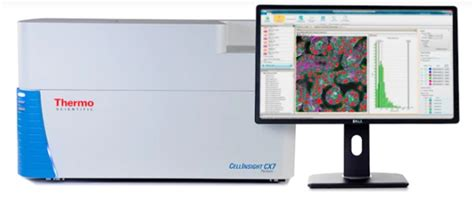
FACS & CellInsigth CX7
For the control of pluripotency maintenance, the platform uses a FACS (Miltenyi) or the automated microscope CellInsigth CX7 (Cellomics ThermoFisher Scentific).