Our History
I-Stem was born in 2005 from the joint desire of the AFM-Telethon and INSERM to create the material and human conditions necessary for French scientists to make up for the delay in exploring the therapeutic potential of human embryonic stem cells caused by the 1994 Bioethics Law, which prohibited all research on the human embryo.
While American and European teams had been working on this since 1998, it was not until August 2004 that the law was modified in France to allow this research.
I-Stem had been in gestation for more than a year in anticipation of this, with the founding idea that scientific forces should be concentrated, geographically and on a well-defined theme. Faced with the recognized irrecoverable delay on the biological mechanisms characteristic of these cells, the choice was made, in agreement with the two supervisory bodies, to move immediately to the next stage, the therapeutic application, and to specifically target genetic diseases. The name of I-Stem naturally followed from this translational ambition (which many people thought was completely out of reach at the time…), thus adding to the “Stem Cell Institute” a very characteristic identity “for the Treatment and Study of Monogenic Diseases”.
The adventure that has now lasted for 18 years thus emerged from the convergence of the scientific bet of a small group of researchers supported by their supervisory authority and the fierce desire of the members of the AFM-Telethon to promote all research likely to bring solutions to patients.
Virtual Tour English – I-Stem, Paris from Amelie Peschanski.
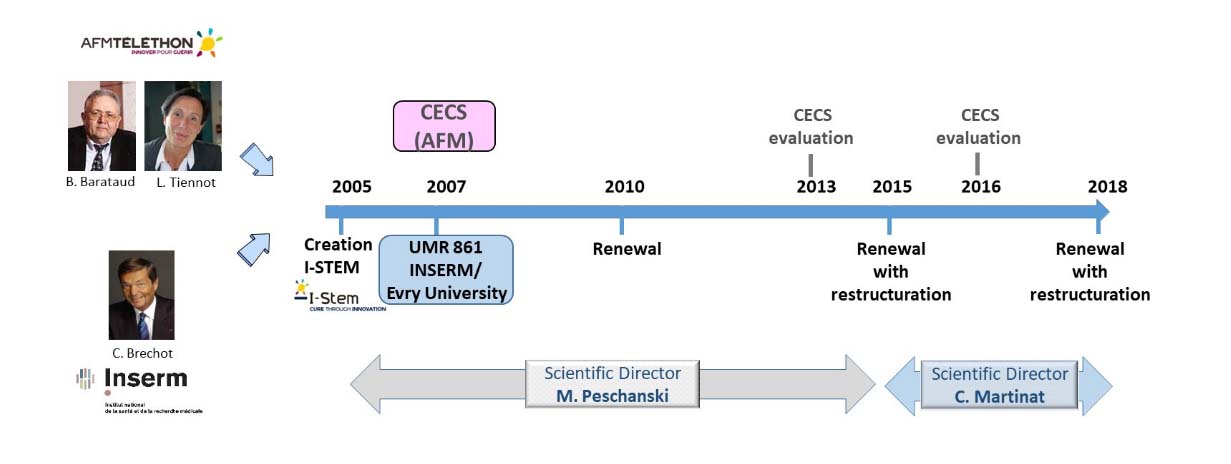
After a first plan written in 2004 that set the framework for a 2-year phase (2005-2006) during which we carried out the feasibility tests prior to building the Institute, the second phase was prepared by a strategic plan drawn up at the end of 2006.
It lasted three years (2007-2009) and was marked by the growth, structuring and demonstration of success of pilot programs.
Phase III, which responded to a strategic plan drawn up at the end of 2009 and lasted until 2015, was a stabilization stage, allowing the way for the realization of clinical expectations, the expansion of pathological indications and the industrialization of processes.
Phase IV, based on a strategic plan drawn up for the period 2016-2020, has as its main objective to provide clinical proof of the concepts we have been developing since the beginning, in regenerative medicine and targeted pharmacology.
After a complex period from 2020 to 2022 due to the pandemic, we are entering a new stage of our strategic development, Phase V: pre-clinical and clinical acceleration & integration of 3D models.
Phase I, 2005 - 2006
Launching period, formation of the team and opening of the laboratory
The first team, made up of some twenty researchers, corresponded to the first phase, the “launch” phase, which lasted two years. It was financed by four partners, three of which were institutionally associated (Inserm, UEVE, AFM) and the fourth (Genopole) which supported the operation. For two years, we assembled a “commando team” to answer three essential questions and define the main principles on which the Institute to be built would be based.
© Yann Arthus Bertrand / projet « Les Français et ceux qui vivent en France ».
Phase II, 2007 - 2009
Growth and demonstration of pilot successes
uring 2006, the positive evaluations of Inserm and AFM allowed the creation of the two administrative entities present until now at I-Stem, the Inserm Unit 861 (mixed with the University of Evry, UEVE) and the Centre d’Etude des Cellules Souches (CECS). These two structures were positively re-evaluated at the end of phase II, in a calendar consistent with the elaboration of the Institute’s third strategic plan.
Phase II was marked by a very strong growth in biological resources, equipment, number of teams and programs.
Phase III, 2010-2015
Focus on therapeutic application programs
From 2010 onwards, I-Stem’s development has continued on the basis of a stabilized workforce (between 70 and 80 employees) reached in 2009. The priority objective put forward in this strategic plan was the preparation of the transition to clinical trials of the results obtained in pre-clinical studies by I-Stem teams. This was therefore precisely a “translational research” phase.
Phase IV, 2016 - 2021
Towards clinical proof of concept validating our pharmacological and regenerative medicine approaches
I-Stem’s 4th strategic plan coincided with the migration of the Institute to brand new premises offered by the Genopole as part of the CRCT (Centre de Recherche Clinique et Translationnelle). We occupy 1600 square meters of space, half of which is divided into four experimental areas of 200 square meters each, devoted respectively to human cell culture (L2 confined laboratories), “dry” biology (biochemistry and molecular biology), robotic production and analysis technologies, and support areas (microscopy, cryopreservation, servers, etc.). The other half of the laboratory is occupied by offices (80 people), the store and the living and circulation areas. There is also access to a large conference room of 200 square meters and several meeting rooms.
Phase V, 2022 - 2027
Clinical and pre-clinical acceleration & integration of 3D models
Après une période complexe de 2020 à 2022 pour raison de pandémie, comme pour tout le monde, nous abordons une nouvelle étape de notre développement stratégique.