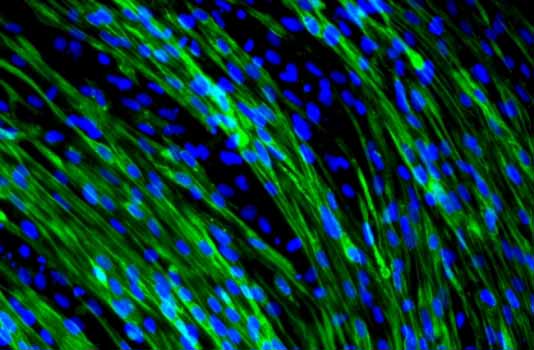
Pharmacology of muscular dystrophies
Our group focuses on using pluripotent stem cells to understand and treat rare genetic disorders. Our work is divided into three axes: pathological modeling, high-throughput pharmacological screening, and the development of innovative approaches based on artificial intelligence.
Rationale :
One of the major advantages of iPS cell technology is that it allows the generation of a potentially unlimited library of human cells suitable for pharmacological applications. While targeting different diseases (HGPS, LGMDs, and GSD III), our experimental paradigm remains the same across all our programs. Following our previous findings and having access to highly reproducible protocols of differentiation, we have embarked on developing drug-repurposing approaches for several diseases by elaborating strategies that can be adapted to each specific situation.
- Identify new molecular mechanisms and biomarkers in iPS cells derivatives
- Develop drug-repurposing approaches targeting pathological phenotypes
- Develop pharmacological approaches targeting proteostasis pathways
- Identify drug candidates through hypothesis-driven or high-throughput screening
Hutchinson Gilford progeria syndrome (HGPS): |
Limb Girdle Muscular Dystrophies (LGMDs): |
Glycogen storage diseases (GSD): |
|---|---|---|
|
Our research started in 2010 with the aim of developing innovative pharmacological approaches for the treatment of HGPS, a rare congenital disease characterized by accelerated aging in children. This syndrome is typically caused by a de novo mutation in the LMNA gene that encodes the major components of the nuclear lamina, the lamins A and C (for review Lo Cicero et al, Ageing Res Rev. 2015). In HGPS, the LMNA mutation results in the production and accumulation of an abnormally farnesylated form of lamin A called progerin (for review Guilbert SM et al, Methods 2021). In 2010, in collaboration with Dr Nicolas Lévy, I-Stem pioneered the generation of several iPS cell lines carrying the C1804T mutation in LMNA to explore the potential of this unique biological resource. From 2010-2018 our group has successfully demonstrated that it was possible to use iPS cell derivatives to recapitulate key features of premature aging in bone (Blondel S 2014 Stem Cells Transl Med. 2014: Lo Cicero A, Sci Rep 2016 Retinoic), skin (Lo cicero et al Sci Rep 2018), and vascular tissues (Ribas J et al Small 2017; Pitrez PR et al ACS Biomater Sci Eng. 2018). We also identified new molecular mechanisms of HGPS, such as the role of miR-9 in the regulation of progerin expression in neurons (Nissan X et al., Cell Reports, 2012). Finally during this period, we also identified several new drug candidates through hypothesis-driven research, such as the positive effect on LMNA alternative splicing of Metformin (Egesipe AL, NPJ Aging Mech Dis. 2016) and MG132 (Harhouri K, et al EMBO Mol Med. 2017) or by high-throughput screening as revealed with the identification of Mono-Aminopyrimidins (Blondel S et al Cell Death Dis 2016) or retinoids (Lo Cicero A, et al Sci Rep 2016). Based on these findings, since 2018, we have pursued our research activity on HGPS in different directions in collaboration with the groups of Nicolas Lévy, Antoine Muchir, Claudia Cavadas and Lino Ferreira identifying pathological mechanisms causing vascular aging in HGPS (Ribas J, et al, Small 2017; Estronca L et al, ACS Biomater Sci Eng. 2018) or new therapetuical options targeting MMP13 (Pitrez PR et al, Nat Commun. 2020) or NAD metabolism (Cardoso et al, submitted). More recently, we have initiated an innovative AI based approach targeting progerin with PROTACs with the Belgium company Kantify. |
In collaboration with Isabelle Richard’s group at Généthon, we have expanded our research to include limb-girdle muscular dystrophies (LGMDs), which are characterized by involvement of the hip and shoulder muscles, progressive muscle degeneration, and increased fibrosis and inflammation. In our team, we use muscle cells derived from induced pluripotent stem cells (iPSCs) to model LGMDs. We have demonstrated that these models reproduce key characteristics of various LGMDs (Bruge et al. 2022) and serve as a robust resource for exploring their molecular mechanisms. We then leverage these models to identify pharmacological treatments using high-throughput screening and drug repurposing approaches (Hoch et al. 2019 Scientific Reports; Hoch et al. 2022 Frontiers in Pharmacology). Among the broad spectrum of LGMDs, we have a particular interest in LGMD R2 (dysferlinopathy). Led by Céline Bruge, this program relies on patient-derived iPS cells to better understand the mechanisms involved in muscle damage and identify effective pharmacological treatments. This research has recently highlighted the potential to repurpose Bazedoxifene as a therapeutic approach (Bruge et al. 2025 British Journal of Pharmacology). At the same time, it is now established that some patients with dysferlinopathies exhibit a slower progression and a later onset of symptoms. These differences suggest the existence of compensatory mechanisms. To identify them, we are currently analyzing muscle biopsies from patients at different stages using omics approaches to highlight protective mechanisms and open up new therapeutic avenues. This project is ongoing. Finally, our recent work has identified DAB2 as a biomarker for LGMDR2 (Bruge et al. 2026 JCI Insight). Beyond its value for diagnosis and monitoring, our results suggest that DAB2 may also contribute to the pathophysiology. DAB2 is also dysregulated in several other muscular dystrophies, revealing mechanisms common to these conditions. This discovery now allows us to explore shared molecular pathways, with the goal of identifying a therapeutic molecule applicable to multiple muscular dystrophies. |
Glycogen storage diseases (GSD) are rare autosomal recessive disorders caused by mutations in genes encoding individual enzymes of the glycogen metabolism pathway. Among them, glycogen storage disease type III (GSDIII; incidence: 1/100 000) is caused by mutations in the AGL gene encoding the glycogen debranching enzyme (GDE) and leads to the accumulation of cytosolic glycogen in the whole body. Although GDE is expressed in all the tissues, the disease manifestation is primarily due to its absence in liver and muscles. During childhood, the disease is mainly metabolic, with hepatomegaly and severe fasting hypoglycemia. In the adulthood, the metabolic disease becomes less prominent and a generalized muscle weakness appears. To date, no treatment is available for GSDIII. Patients follow strict diet regimens to avoid recurrent hypoglycemia. Started in 2020 in collaboration with Giuseppe Ronzitti‘s group at Généthon this program was recently awarded by an ANR JCJC, a grant from the Foundation Maladies Rares and the support of the Association Francophone des Glycogénoses. Led by Lucile Hoch this program aims to use patients derived pluripotent stem cells to understand the molecular mechanisms involved in the muscular pathophysiology of the disease and identify an efficient pharmacological treatment to clear glycogen accumulation in muscles. |
DREAMS Consortium :
The DREAMS consortium is a European consortium whose objective is to reposition drugs for muscular dystrophies through the cross-utilization of pluripotent stem cells and artificial intelligence. Coordinated by Xavier NISSAN, the consortium is composed of nine partners.
1 – Xavier Nissan (CECS) Coordinateur
2 – Nik Subramanian, Segolene Martin (KANTIFY) co-coordinateur
3 – Lino Ferreira (CNC)
4 – Stephane Vassilopoulos et Antoine Muchir (Institut de myologie)
5 – Karim Whabi et Teresinha Evangelista (APHP)
6 – Shenhav Cohen (TECHNION)
7 – John Blackwood Peter Hamley (SAMSARA)
8 – Alexandre Mejat (AFM-Téléthon)
9 – ZABALA
Team members
Xavier NISSAN
Research Director
Xavier joined the laboratory in 2005 to develop pharmacological approaches for the treatment of genetic diseases. His expertise is focused on the establishment of preclinical models and the development of pharmacological screenings.
Céline BRUGE
Researcher
Since her arrival at I-Stem in 2016, Celine has been interested in dysferlinopathies. Her research aims to identify new biomarkers and to identify pharmacological agents able to regulate them.
Quentin Miagoux
Bioinformatics Research Engineer
Bio-informatician by training, Quentin is interested in omics and screening data analysis. As part of a partnership with Kantify, he is looking to use AI to identify new therapeutic targets for genetic diseases. Quentin holds a PhD in Life Sciences and Health / Bioinformatics.
Manon BENABIDES
Engineer
Manon works on the LGMD R9 pathology. She is looking for treatments for this disease through modeling and phenotypic screening approaches. She is also the lab manager of the team. Manon holds a Master’s degree in Life Sciences, specializing in biotechnology and cellular engineering.
Emilie PELLIER
Engineer
Emilie is working on the pathophysiological consequences caused by the absence of dysferlin in LGMD R2. She is particularly interested in the membrane repair capacity of muscle cells. Emilie graduated with a Master’s degree in Bio-Health / Tissue, Cellular and Gene Biotherapies (BTCG).
Matthieu Lejars
Research assistant (CECS)
Matthieu holds a Master’s degree in Translational Immunology and Biotherapies, and joined the team in November 2023 to work on the DREAMS project. He is involved in the development of pluripotent stem cells of interest, as well as their differentiation into muscle progenitors.
Juliette Lemoine
Research Associate
Juliette, who holds a PhD in life sciences and specializes in gene therapy, joined the team in September 2025 as a postdoctoral researcher to contribute to the DREAMS project. She is involved in setting up a high-throughput screening based on autophagy and focuses her work on Danon disease.
Hassan Hayat
Phd Student
Trained as a pharmacist and holder of a Master’s degree in Medicines and Health Products specialized in innovative therapies, Hassan joined the team in February 2025 as an intern within the DREAMS project. Awarded a fellowship from the SDSV Doctoral School (Université Paris-Saclay), he began his PhD in October 2025, aiming to identify regulatory hubs common to muscular dystrophies using a multi-omics approach.
Perrine Tennevet
Research Assistant (CECS)
Collaborations
Publications
DAB2 in LGMD R2: a molecular link between disease progression and lipid dysregulation.
23 March 2026
JCI insight
Basket trials in rare diseases: a systematic review of current practices, methodological challenges, and future directions.
12 November 2025
Orphanet journal of rare diseases
Generation and characterization of three human induced pluripotent stem cell lines from patients with glycogen storage disease type II.
01 October 2025
Stem cell research